
Yet, initial ADCs pairing standard anti-cancer agents, such as doxorubicin, were ineffective in clinical trials ( 1). Such a combination can potentially minimize dose-limiting toxicities while maximizing desired therapeutic effects. Establishing a set of selection and analytical criteria will facilitate the translation of novel ADCs and ensure the production of effective biosimilars.Īntibody drug conjugates (ADCs) couple the highly desirable pharmacokinetic (PK) profile and targetability of monoclonal antibodies (mAbs) with the potent cytotoxicity of small molecule drugs. 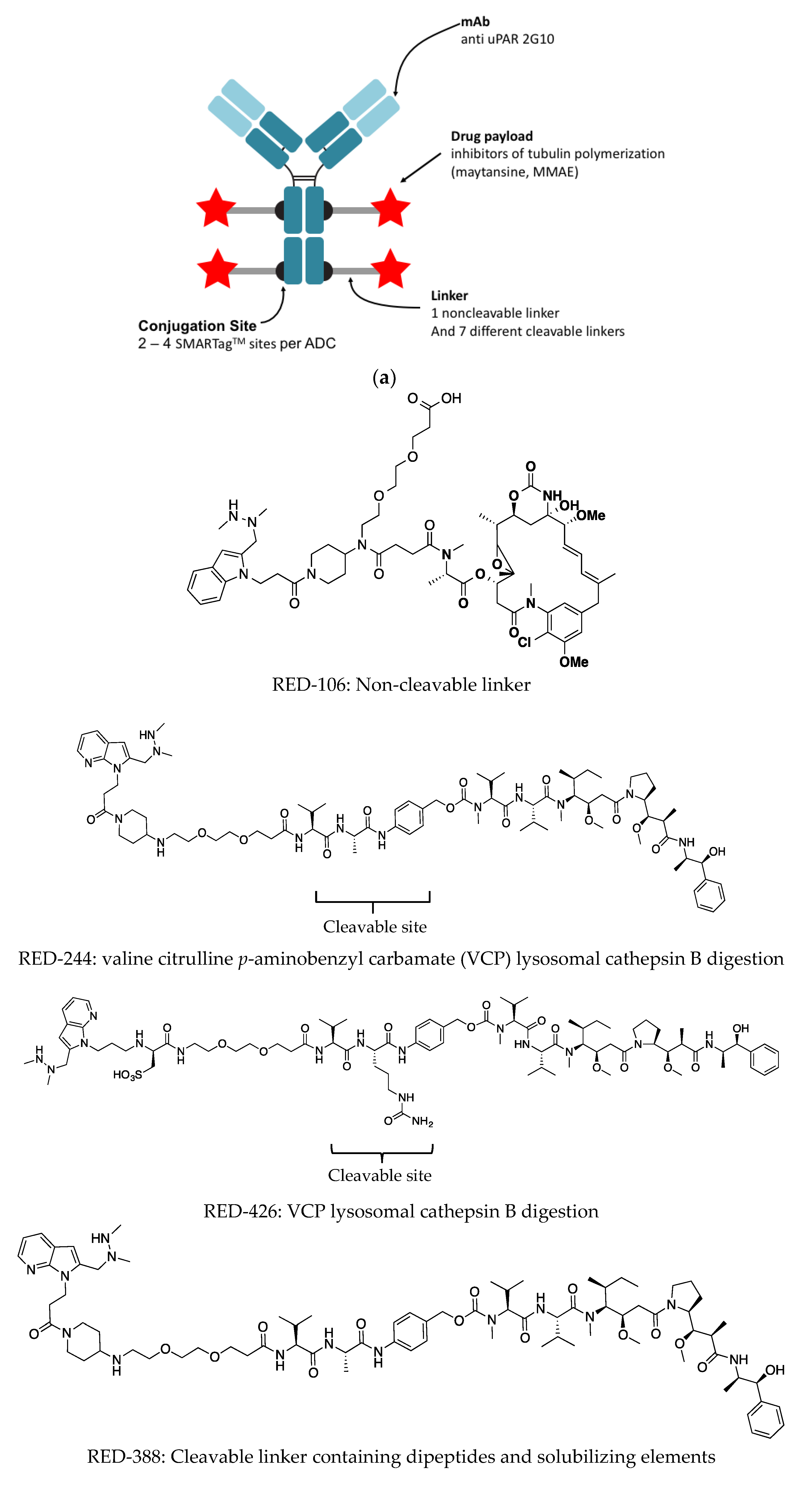
In addition, we report on the progress in characterizing to determine physicochemical properties and on advances in purifying to obtain homogenous products. We discuss current linker technology, contrasting the characteristics of cleavable and non-cleavable linkers, and summarize the essential properties of ADC payload, centering on chemotherapeutics. In this article, we review popular conjugation methods and highlight recent approaches including “click” conjugation and enzymatic ligation. Recently, considerable attention has been focused on developing methods to control the site and number of linker/drug conjugated to the antibody, with the aim of producing more homogenous ADCs. The three main components of ADCs are the antibody, the linker, and the payload the majority of early work focused intensely on improving the functionality of these pieces. 
Antibody drug conjugates (ADCs) have emerged as an important pharmaceutical class of drugs designed to harness the specificity of antibodies with the potency of small molecule therapeutics.
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
